Treatment of severe infections caused by pathogenic bacteria relies on ‘last resort’ antibiotics, but rising resistance by ‘superbugs’ to most clinically approved drugs leaves patients exposed to possible fatalities.
Flinders University researchers are focusing on how bacterial cells adapt and resist antimicrobial medications – with a new article focusing on hospital strain of Acinetobacter baumannii and its cellular response to important antibiotic colistin.
The World Health Organization names antibiotic resistance as one of the biggest threats to global health, food security, and development with a growing number of infections – including pneumonia, tuberculosis, gonorrhoea and salmonellosis – becoming harder to treat as antibiotics used to treat them become less effective.
Antibiotic resistance leads to longer hospital stays, higher medical costs and increased mortality, researchers warn.

“Around the world, there are fewer and fewer new antibiotics being identified and produced for medical use – and this is compounded by the ever-increasing antibiotic resistance seen in bacterial strains causing infections,” says Flinders microbial researcher Dr Sarah Giles.
“If we can understand the bacterial mechanisms, such as this, we can potentially apply new therapies to treat patients – particularly those with multi-drug-resistant bacterial infections.”
“We noted that the A. baumannii bacterial strain had a two-part signal system which altered its potential response to antibiotic treatment,” says Dr Sarah Giles, as part of a NHMRC-Flinders University Research Scholarship study.
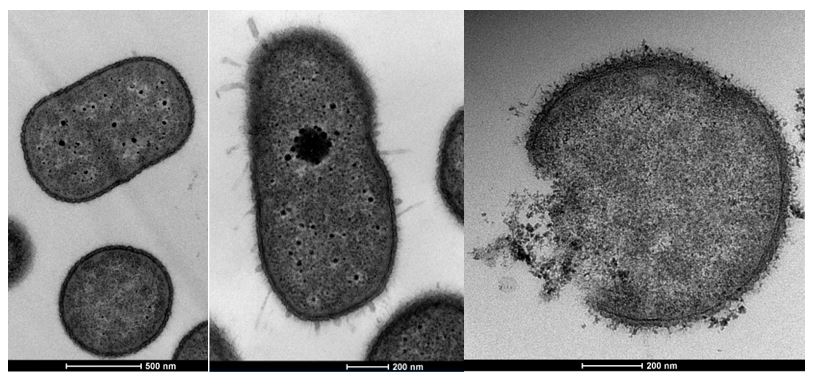
This ‘two-component signal transduction’ observed involves a response regulator protein in the StkR/S system acting as a repressor and when genetically removed hundreds of transcriptional changes are seen.
Some of these changes affect the bacterial cell’s outer membrane composition leading to colistin resistance.
“Colistin is known as a ‘last resort’ antibiotic and therefore identifying and understanding the mechanisms contributing to bacterial antibiotic-resistant is critical,” says senior researcher Professor Melissa Brown.
Antimicrobial resistance (AMR) occurs when bacteria, viruses, fungi and parasites change over time and no longer respond to medicines, making infections harder to treat and increasing the risk of disease spread, severe illness and even death.
The article, The StkSR Two-Component System Influences Colistin Resistance in Acinetobacter baumannii (May 2022), by Sarah K Giles, Uwe H Stroeher, Bhavya Papudeshi, Rob Edwards, Jessica AP Carlson-Jones, Michael Roach and Melissa H Brown has been published in the Microorganisims DOI: 10.3390/microorganisms10050985
Acknowledgement: The study was funded by the National Health and Medical Research Council (grant 535053) to Professor Melissa Brown, and a Flinders University Research Scholarship to Dr Sarah Giles.

