
Revolutionary ‘green’ types of bricks and construction materials could be made from recycled PVC, waste plant fibres or sand with the help of a remarkable new kind of rubber polymer discovered by Australian scientists.
The rubber polymer, itself made from sulfur and canola oil, can be compressed and heated with fillers to create construction materials of the future, says a new paper unveiling a promising new technique just published in Chemistry – A European Journal.
“This method could produce materials that may one day replace non-recyclable construction materials, bricks and even concrete replacement,” says organic chemistry researcher Flinders University Associate Professor Justin Chalker.
The powdered rubber can potentially be used as tubing, rubber coatings or bumpers, or compressed, heated then mixed with other fillers to form entirely new composites, including more sustainable building blocks, concrete replacement or insulation.
Cement is a finite resource and heavily polluting in its production, with concrete production estimated to contribute more than 8% of global greenhouse gases emissions, and the construction industry worldwide accounting for about 18%.
“This new recycling method and new composites are an important step forward in making sustainable construction materials, and the rubber material can be repeatedly ground up and recycled,” says lead author Flinders PhD Nic Lundquist. “The rubber particles also can be first used to purify water and then repurposed into a rubber mat or tubing.”
“This is also important because there are currently few methods to recycle PVC or carbon fibre,” he says, with other collaborators from Flinders University, Deakin University and University of Western Australia.

Co-author and research collaborator Dr Louisa Esdaile says the important research looks at ways to repurpose and recycle materials, so that these materials are multi-use by design.
“Such technology is important in a circular economy,” says Dr Esdaile, a special contributor to this month’s Young Chemist issue of Chemistry – A European Journal (ChemEurJ).
The new manufacturing and recycling technique, labelled ‘reactive compression molding,’ applies to rubber material that can be compressed and stretched, but one that doesn’t melt. The unique chemical structure of the sulfur backbone in the novel rubber allows for multiple pieces of the rubber to bond together.
The project started two years ago in the Chalker Laboratory as a third-year project by Ryan Shapter, with Flinders University PhD candidates Nicholas Lundquist and Alfrets Tikoalu and others contributing to the paper in this month’s special Young Chemist issue of ChemEurJ.
‘Reactive compression molding post‐inverse vulcanization: A method to assemble, recycle, and repurpose sulfur polymers and composites’ (May 2020) by N Lundquist, A Tikoalu, M Worthington, R Shapter, S Tonkin, F Stojcevski, M Mann, C Gibson, J Gascooke, A Karton, L Henderson, L Esdaile, JM Chalker in Chemistry – A European Journal. DOI: 10.1002/chem.202001841
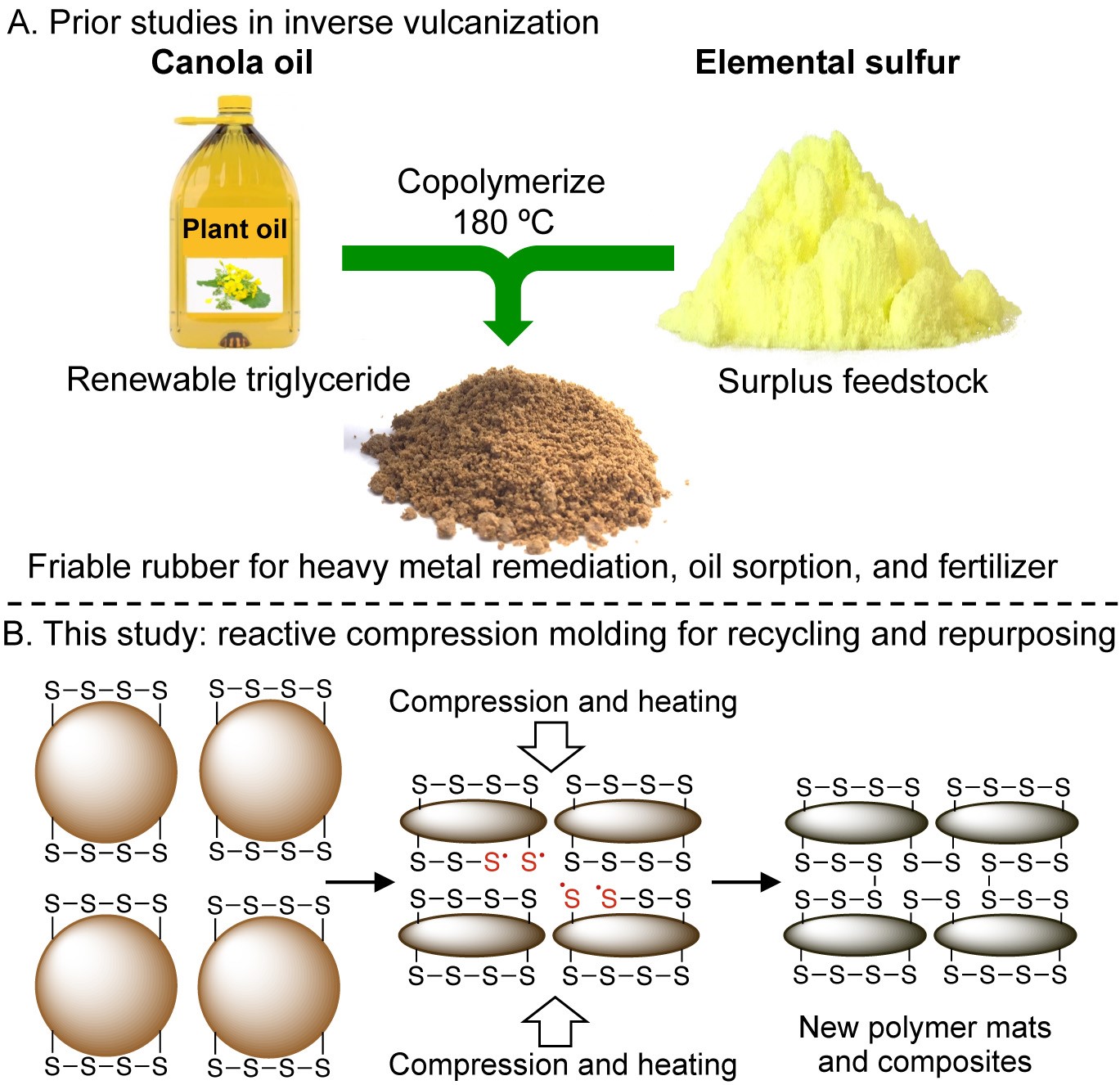 The Chalker Lab at Flinders University has previously developed sulfur polymers for a number of applications in mopping up environmental pollutants – from oil spills in water and mercury and heavy metals in soil.
The Chalker Lab at Flinders University has previously developed sulfur polymers for a number of applications in mopping up environmental pollutants – from oil spills in water and mercury and heavy metals in soil.
Computational studies led by collaborators at UWA also revealed fundamental insight into how the length of the sulfur chains in the rubber is important for reaction compression molding. The project was also collaboration with the Institute for Frontier Materials at Deakin University.

